Gene editing? CRISPR? Everyone has heard the term CRISPR from mainstream media. But few can explain what it is or its significance for the future of human medicine. Hailed as a 21st century miracle, Jennifer Doudna and Emmanuelle Charpentier were recently awarded the Nobel Prize for their breakthrough research on CRISPR technology in December 2020. But the discovery of CRISPR actually dates as far back as the 1980’s. Scientists just had no idea what they were looking at except that some bacteria carried strange repeating bits of DNA which was eventually given an acronym CRISPR (clustered regularly interspaced short palindromic repeats).
What is the significance of these repeating patterns of DNA in bacteria? Just like humans, bacteria are also attacked by viruses. To fight against future viral infections these bacteria have evolved their own immune system through a CRISPR region in their DNA, which encodes molecules that can recognize viral genes and cut them before they invade the bacterial host. Scientists have figured out how to harness this natural genetic engineering to artificially create CRISPR molecules that can snip out pieces of genes, swap in a new piece of DNA, which can then stitch itself back together. The fact that CRISPR exists in nature, is cheap to make, is so accurate due to years of natural genetic evolution and requires little effort on the part of the researchers is what gives it a potential advantage over previous gene editing technologies conceived in labs such as TALEN (transcription activator-like effector nucleases) and ZFN (zinc finger nucleases). There are also new gene editing approaches emerging such as base editing, prime editing, and even gene writing. Certain companies are also developing proprietary editing technologies that do not necessarily belong to any of the above groups. It is still unclear which is the superior gene editing technology and there are proponents for each but given the number of companies using CRISPR and how it has changed the field, it appears to be the most popular.
Humans are burdened by thousands of different genetic diseases, which are due to inherited mutations. Most small molecule drugs target proteins that result in disease symptoms due to the faulty gene but do not address the actual underlying inherited mutation in the gene. Gene editing offers the possibility to potentially edit such inherited mutations so that patients no longer have to keep taking life-long medications. Sickle cell anemia is an example of a disease currently being studied in the clinic using CRISPR. People with sickle cell anemia inherit two faulty genes for hemoglobin, the oxygen carrying molecule in red blood cells, which results in red blood cells that are shaped like sickles or crescent moons instead of the usual flexible and round shape. These sickled cells can block blood flow and cause pain, organ damage, fatigue, and infection. Sickled cells also tend to die earlier than healthy red blood cells and thus compromise oxygen delivery. Ironically sickle cell anemia is an evolutionary genetic condition that protects against malaria, which ravages sub-Saharan Africa, but results in the very symptoms listed above and reduced life expectancy. Current medications only address symptoms. Stem cell or bone marrow transplants can induce cure but have significant risks associated with the procedure and are rarely done. CRISPR and other gene editing approaches offer a new possibility, whereby the mutation can be erased that causes sickle cell anemia.
Gene editing has so many potential applications beyond just genetic medicine and can be used to make tweaks to DNA of other animals and plants in order to create new breeds of livestock or plants to address potential future food shortages. There is still tremendous regulation when it comes to gene editing. In 2018 a rogue scientist in China (who is currently in prison) used CRISPR on the DNA of human embryos to edit out the gene that codes for a protein that HIV uses to enter cells. Does this forecast an ominous future of designer babies or DNA tinkering to gain an unfair advantage? Only time will tell but a precedent has now been set to reflect on.
CRISPR and other gene editing technologies still require more experiments, research grant applications, investment pitches, regulatory approval, manufacturing agreements, supply chain configurations and so much more, which will take years to navigate, but the future of gene editing as a functional therapy as well as for diagnostics and biomarker discovery is near.
Current leading publicly traded gene editing therapeutics companies:
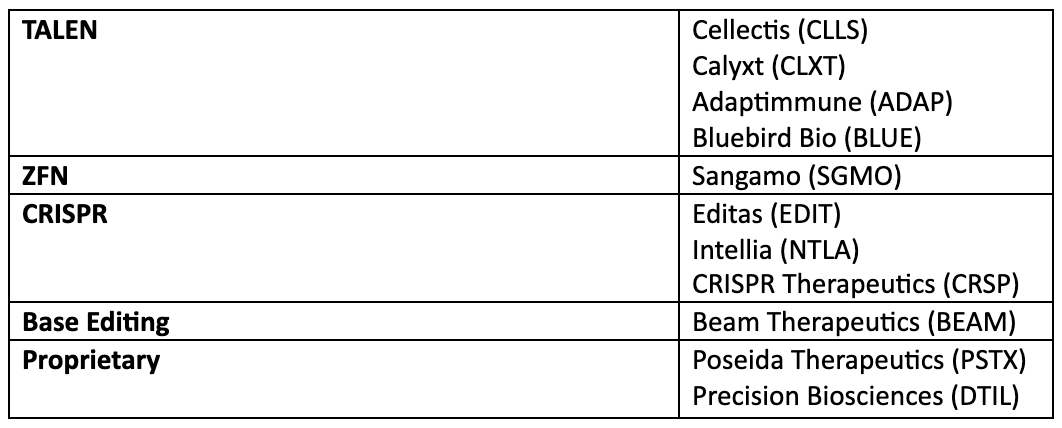
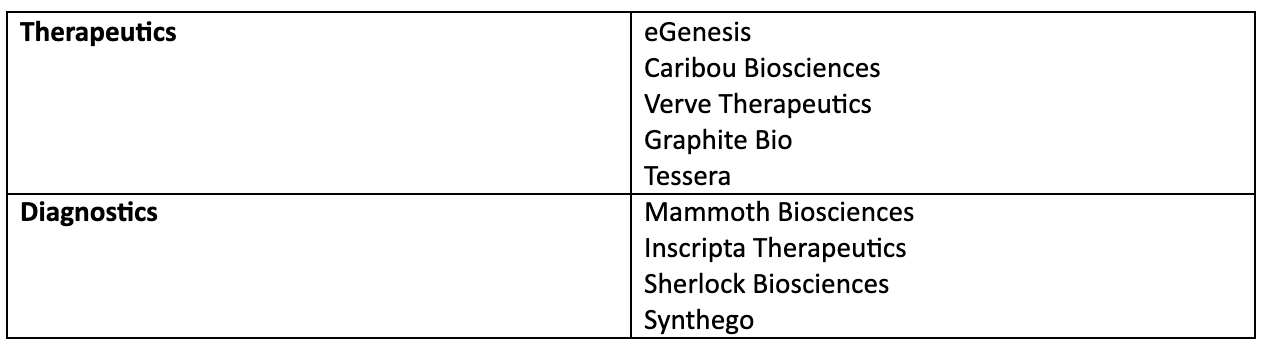
Majority of gene editing companies are still private but have tremendous venture capital investments and will likely IPO in the coming years. Some of the well-known names that are using gene editing for therapeutic or diagnostic purposes are:
Gilmartin has direct expertise within the space and has partnered with many clients to start building an Investor Relations program when private. For additional information, please contact our team today.
Priyam Shah, Principal

Leave a Reply